Shanghai, China, April 23, 2026 — Shanghai Henlius Biotech, Inc. (2696.HK) today announced the first comprehensive presentation of its proprietary antibody-drug conjugate (ADC) platform, Hanjugator™, along with the latest research progress of multiple innovative ADC candidates derived from the platform, at the 2026 Annual Meeting of the American Association for Cancer Research (AACR). The data indicate that by optimizing payload potency and linker design, Hanjugator™ has the potential to expand the therapeutic window while enhancing antibody functionality, offering a novel technological approach for the treatment of solid tumors.
Hanjugator™ ADC Platform: Tunable payloads and a high-efficiency, low-toxicity design to maximize antibody potential and expand the therapeutic window
In the design of ADCs, highly potent cytotoxic payloads can effectively kill tumor cells but often limit clinically tolerable dosing, preventing optimal antibody exposure and resulting in a narrow therapeutic window. To address this challenge, Henlius has developed Hanjugator™, a next-generation, proprietary ADC platform featuring a customizable and modular camptothecin-based linker-payload system. This platform is designed to precisely tune payload potency, thereby maximizing antibody-mediated pathway blockade and improving the overall therapeutic window.
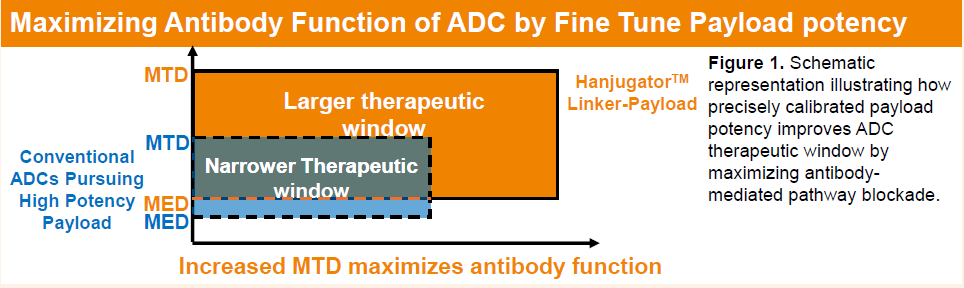
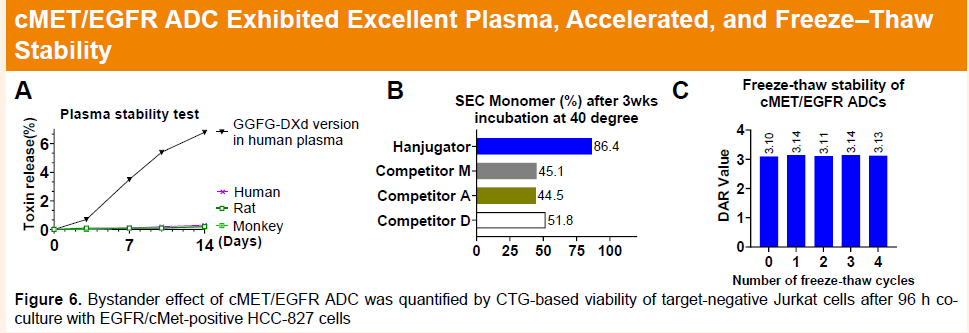
A key advantage of the platform lies in its hydrophilic camptothecin-based linker-payload design, which is compatible with unconventional hydrophobic antibodies and demonstrates strong thermal stability, plasma stability, and freeze–thaw stability. In addition, Hanjugator™ enables flexible modulation of payload potency, allowing selection of optimal cytotoxic strength tailored to specific targets, with activity ranging from approximately one-third that of DXd to levels comparable to exatecan. This design facilitates an optimal balance between antibody-mediated signaling blockade/internalization and small-molecule cytotoxicity, potentially achieving synergistic therapeutic effects and expanding applicability across diverse targets and indications.
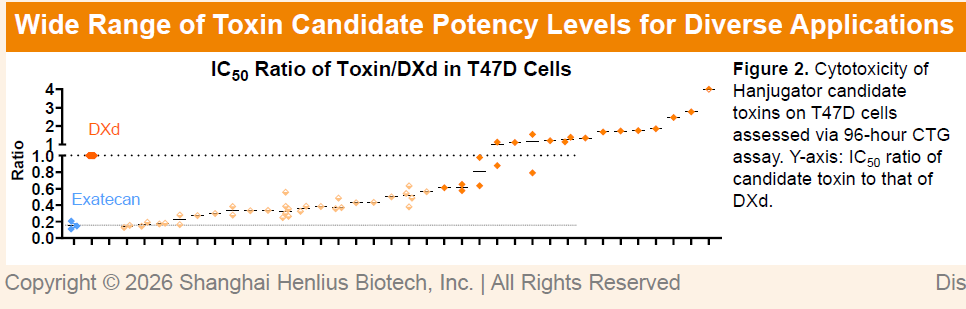
Furthermore, ADCs generated from the Hanjugator™ platform exhibit a bystander killing effect more than tenfold greater than that of deruxtecan-based ADCs, which may help overcome tumor heterogeneity and deliver superior overall efficacy compared with multiple linker-payload systems currently in clinical development. The differentiated payload design may also help mitigate common mechanisms of toxin resistance, providing a novel solution for next-generation ADC development. To date, the platform has generated more than 12 early-stage candidates, including HLX48 (cMET/EGFR bispecific ADC), HLX49 (HER2 biparatopic ADC), HLX403 (CDH17 ADC), HLX402 (ADAM9 ADC), HLX85 (ALPP/ALPPL2 ADC), and HLX41 (LIV-1 ADC).
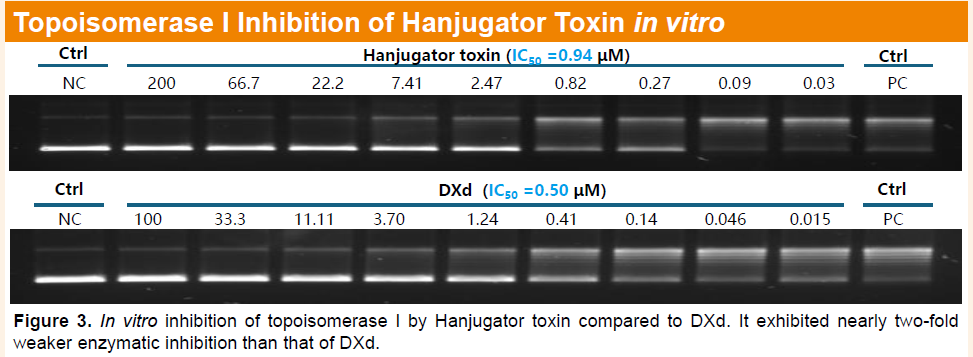
Latest Advances at AACR: First disclosure of preclinical data for lead assets including the cMET/EGFR bispecific ADC HLX48 and the HER2 biparatopic ADC HLX49
HLX48 (cMET/EGFR bispecific ADC)
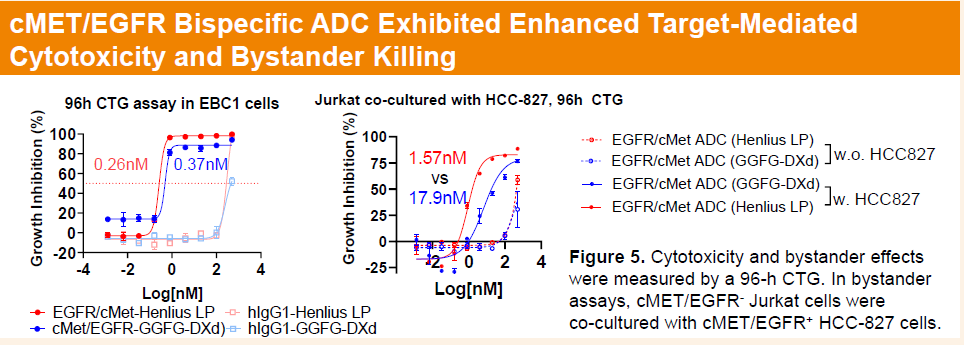
HLX48 is an cMET/EGFR bispecific ADC comprising a dual-targeting antibody conjugated to a camptothecin-based topoisomerase I inhibitor payload. Given that cMET/EGFR bispecific ADCs typically require higher clinical doses to achieve optimal receptor occupancy, HLX48 adopts a hydrophilic linker strategy and a payload with moderate potency, with a drug-to-antibody ratio (DAR) of approximately 4, aiming to balance strong antitumor activity with manageable toxicity. The investigational new drug (IND) application for HLX48 has been accepted by the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) in China and has also been filed with Australia’s Therapeutic Goods Administration (TGA).

-
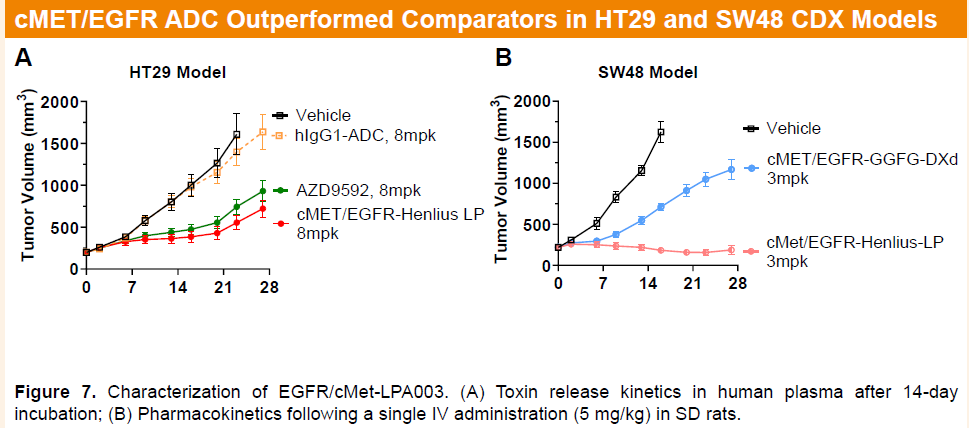
In HT29 and SW48 human colorectal cancer CDX models, HLX48 also showed superior efficacy compared with AZD9592.
-
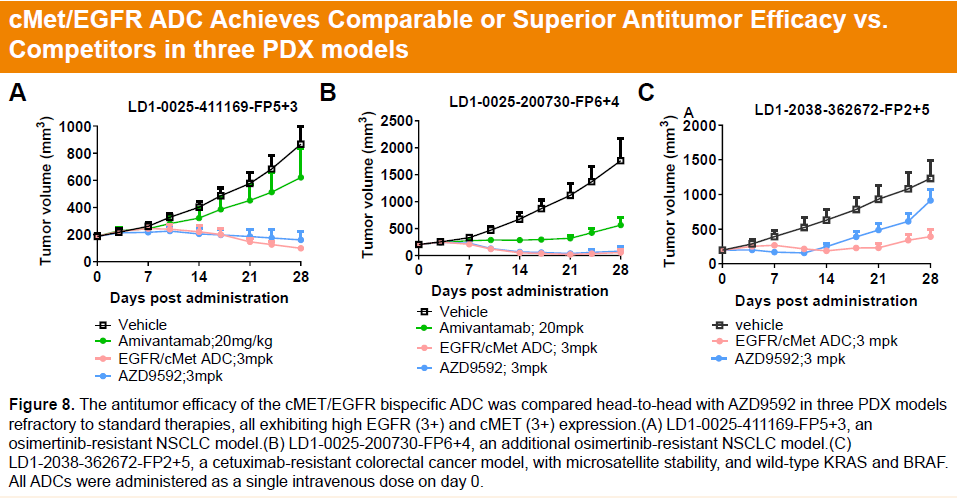
Pharmacology studies further demonstrated that HLX48 achieved comparable or superior antitumor activity relative to benchmark agents across three PDX models resistant to standard therapies, with a single 3 mg/kg dose inducing significant tumor regression.
-
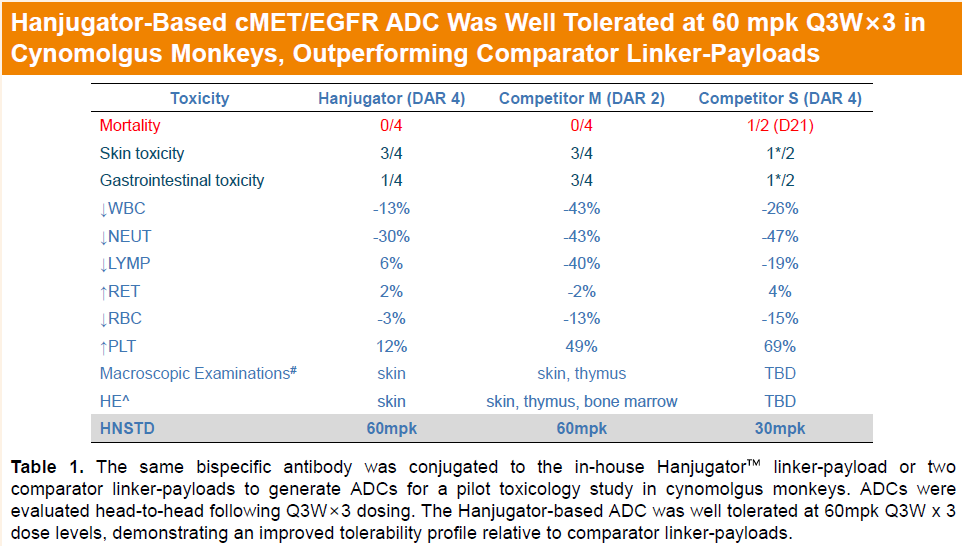
In cynomolgus monkeys, HLX48 was well tolerated at 60 mg/kg under a Q3W×3 dosing regimen, demonstrating improved tolerability relative to comparator linker-payloads and supporting the feasibility of its moderate-potency payload design.
HLX49 (HER2 biparatopic ADC)
HLX49 is a HER2 biparatopic ADC developed based on trastuzumab (HANQUYOU) and a novel anti-HER2 antibody (dulpatatug*), enabling simultaneous binding to distinct HER2 domains. This design enhances internalization of HER2 homodimers and HER2/EGFR heterodimers, resulting in stronger HER2 signaling blockade. With an efficient, low-toxicity payload, HLX49 allows higher dosing and enhanced receptor engagement, thereby enhancing tumor cell killing.
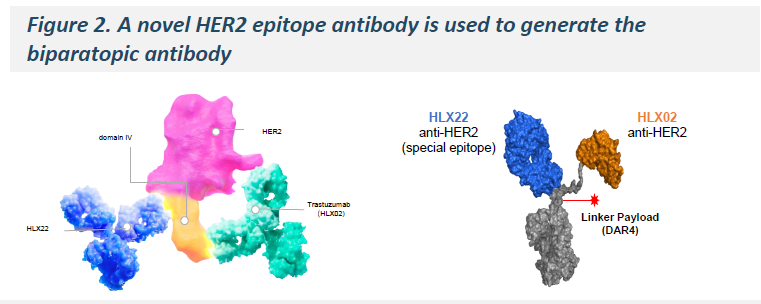
-
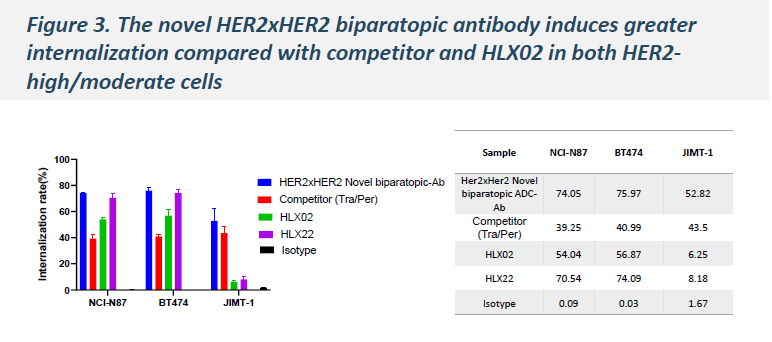
In BT-474 and NCI-N87 cell lines, HLX49 demonstrated superior internalization efficiency compared with KN026 and trastuzumab deruxtecan (T-DXd), and exhibited improved antitumor efficacy across multiple cell lines.
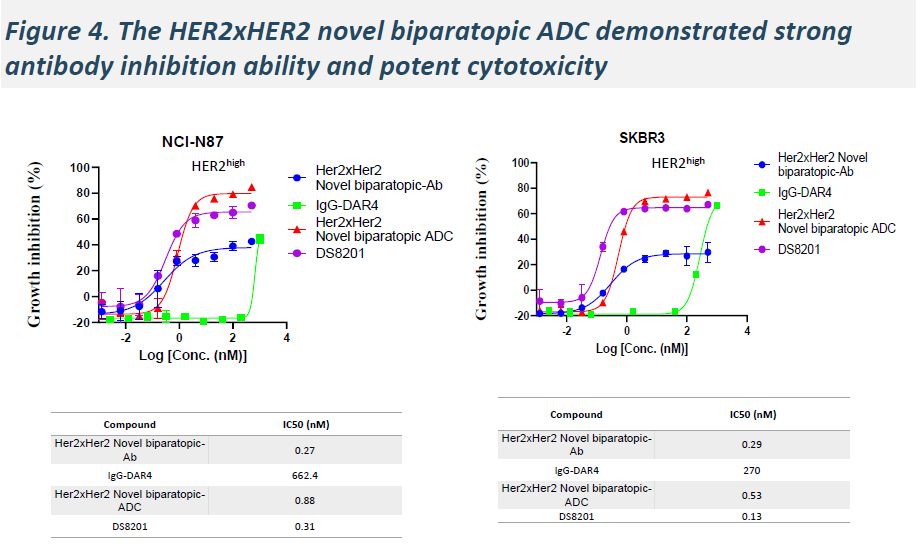
-
In xenograft models spanning HER2-positive, HER2-low, and HER2-ultralow expression levels, a single 6 mg/kg dose of HLX49 induced significant tumor regression and outperformed T-DXd in head-to-head comparisons.
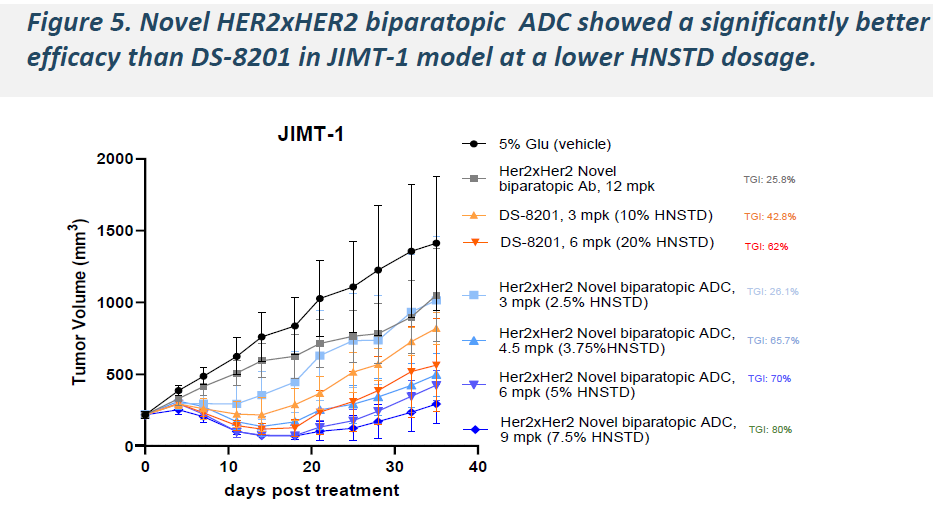
-
Preliminary toxicology studies in cynomolgus monkeys indicated that HLX49 was well tolerated following three doses at 60 mg/kg.
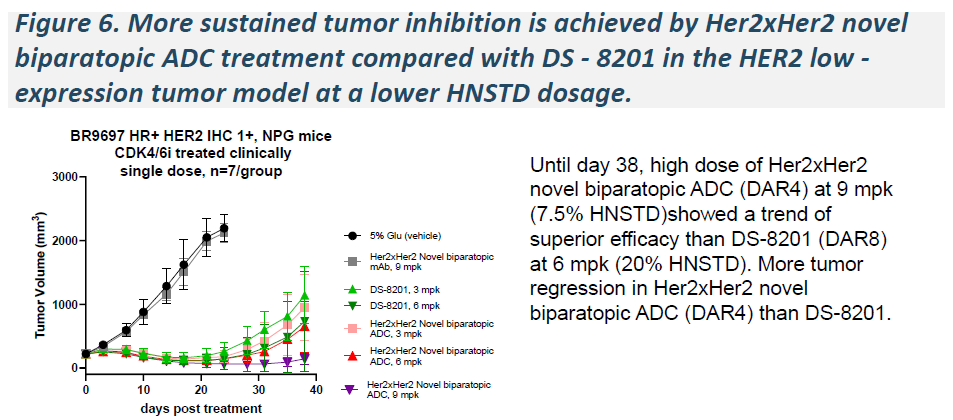
HLX402(ADAM9 ADC)
HLX402 targets ADAM9 (a disintegrin and metalloproteinase 9), a protein involved in proteolytic shedding of cytokines and growth factors and implicated in tumor progression, metastasis, and angiogenesis. While ADAM9 is overexpressed in multiple solid tumors, previous programs targeting this molecule have been limited by ocular toxicity concerns. HLX402 incorporates a novel camptothecin-based linker-payload designed to mitigate such toxicity while expanding the therapeutic window.

-
Preclinical studies showed that the parental anti-ADAM9 antibody binds to human and cynomolgus ADAM9 with nanomolar affinity and exhibits no cross-reactivity with rodent orthologs or other ADAM family members, confirming high target specificity. Relative to a benchmark antibody, the anti-ADAM9 antibody demonstrated superior internalization in Miapaca-2 cells, while the corresponding ADC (HLX402) exhibited comparable or enhanced internalization in CHOK1-hADAM9 cells. Furthermore, HLX402 displayed potent cytotoxicity and pronounced bystander killing activity in vitro.

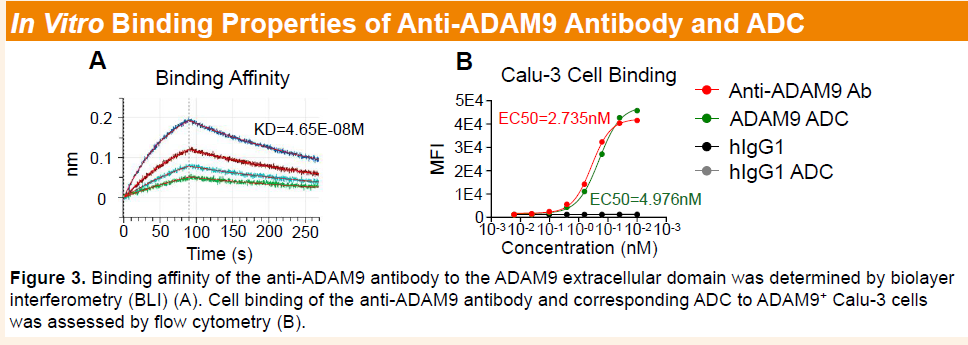
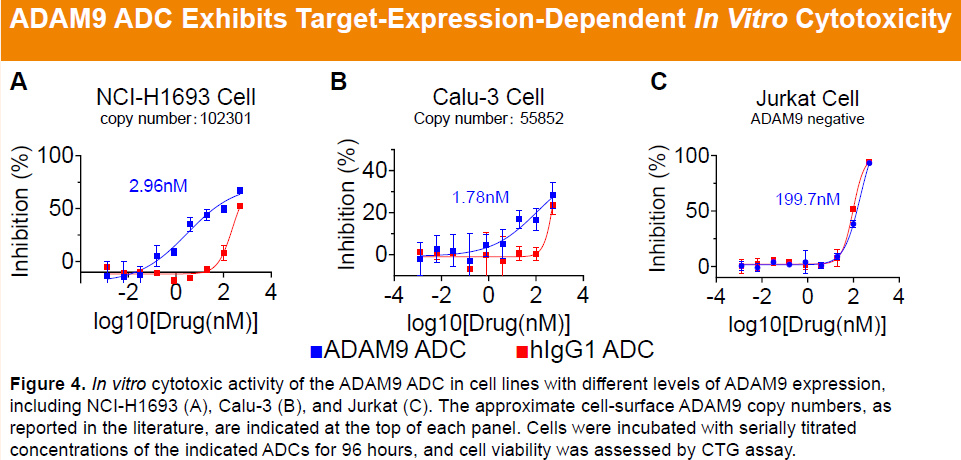

-
In the Calu-3 CDX model, a single dose of 3 mg/kg HLX402 achieved 109.3% tumor growth inhibition (TGI), outperforming the benchmark ADC (91.1%). In the DLD-1 CDX model, HLX402 demonstrated dose-dependent efficacy, with TGIs of 66.8% and 90.2% at 3 mg/kg and 8 mg/kg, respectively, significantly exceeding the benchmark ADC, which achieved 39.2% TGI at 3 mg/kg.
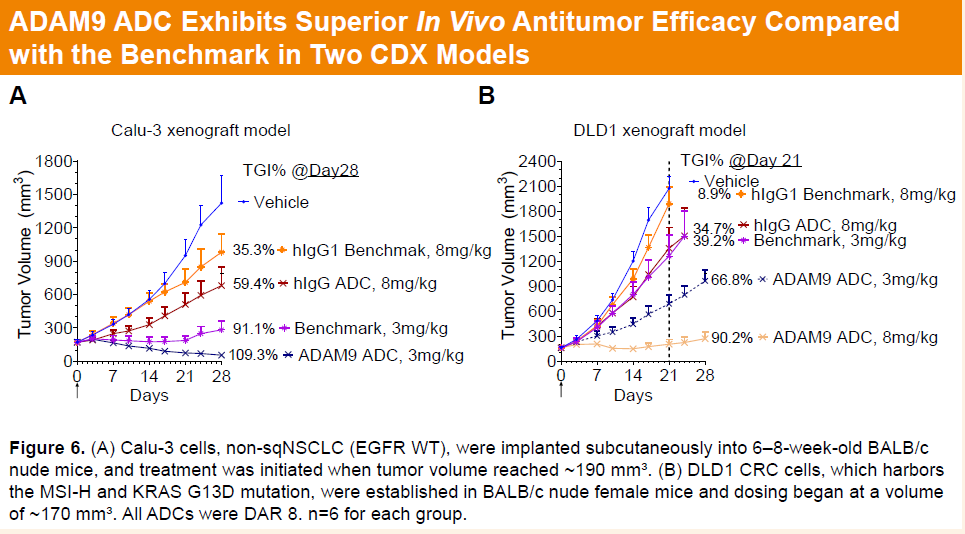
-
HLX402 demonstrated excellent plasma stability, with minimal changes in drug-to-antibody ratio (DAR) and low levels of aggregation observed in both accelerated and freeze–thaw stability studies. In rats, HLX402 exhibited favorable pharmacokinetic properties, characterized by minimal release of free payload and an antibody-like half-life.
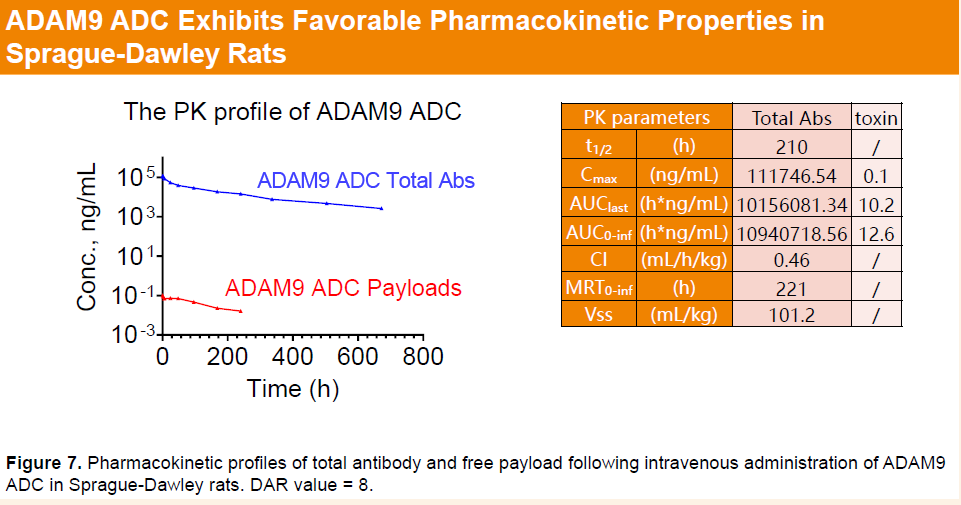
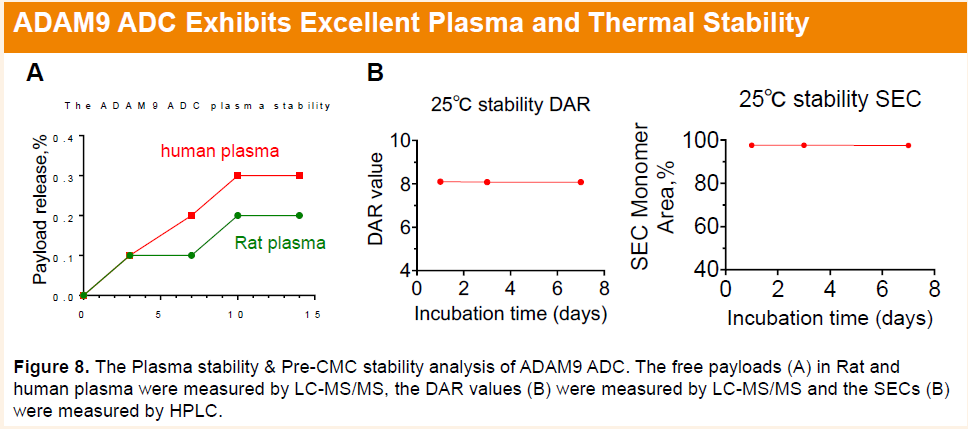
HLX85(ALPP/ALPPL2 ADC)
HLX85 targets placental alkaline phosphatase (ALPP) and placental alkaline phosphatase 2 (ALPPL2), which are highly expressed across multiple solid tumors but show limited expression in normal tissues, making them attractive therapeutic targets. As a potential first-in-class camptothecin-based ADC targeting ALPP/ALPPL2, HLX85 consists of a high-affinity monoclonal antibody conjugated to a proprietary camptothecin payload via a protease-cleavable linker, highlighting its strong development potential in solid tumors, particularly gynecologic malignancies.
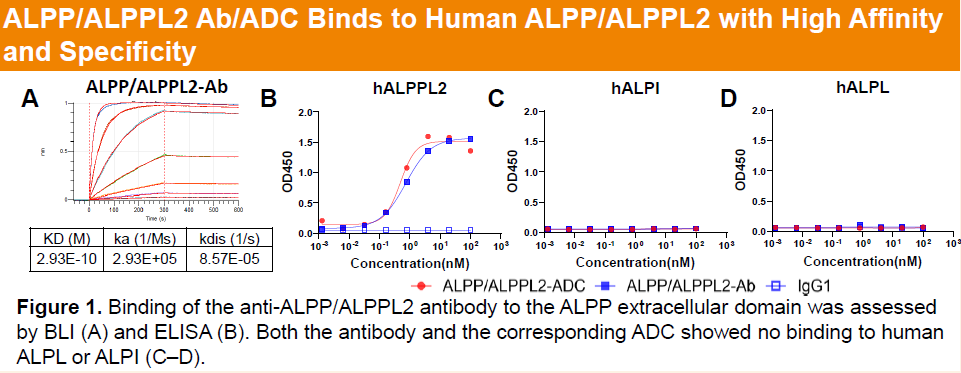
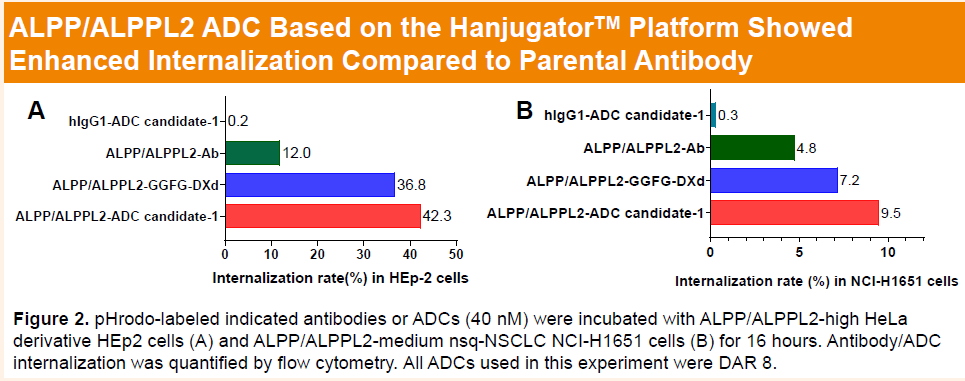
-
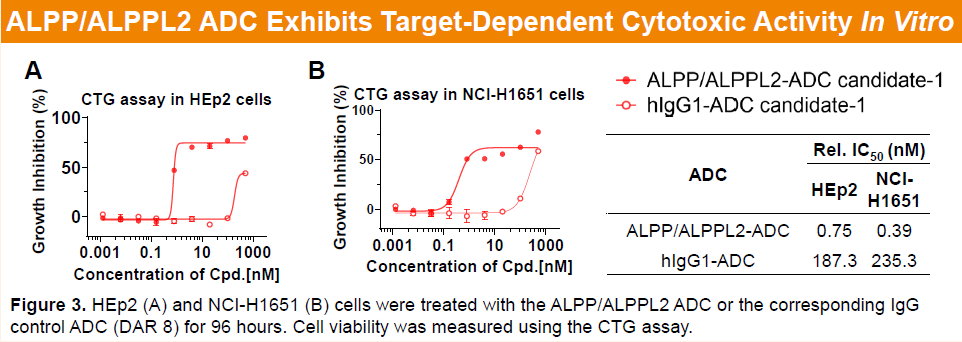
Preclinical studies showed that the parental ALPP/ALPPL2 antibody of HLX85 exhibits high specificity toward ALPP/ALPPL2, with no cross-reactivity to related phosphatases (hALPL and hALPI). HLX85, with a drug-to-antibody ratio (DAR) of approximately 8 and incorporating a proprietary camptothecin-based linker–payload system, demonstrated pronounced target-dependent cytotoxicity across different levels of ALPP/ALPPL2 expression, including HEp2 cells (high expression) and NCI-H1651 cells (moderate expression).

-
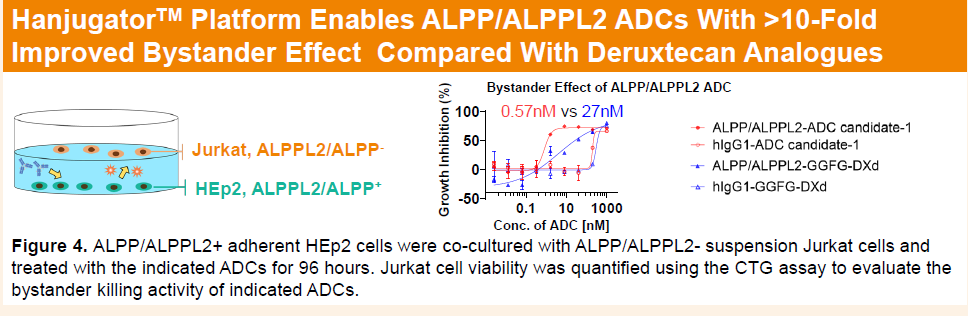
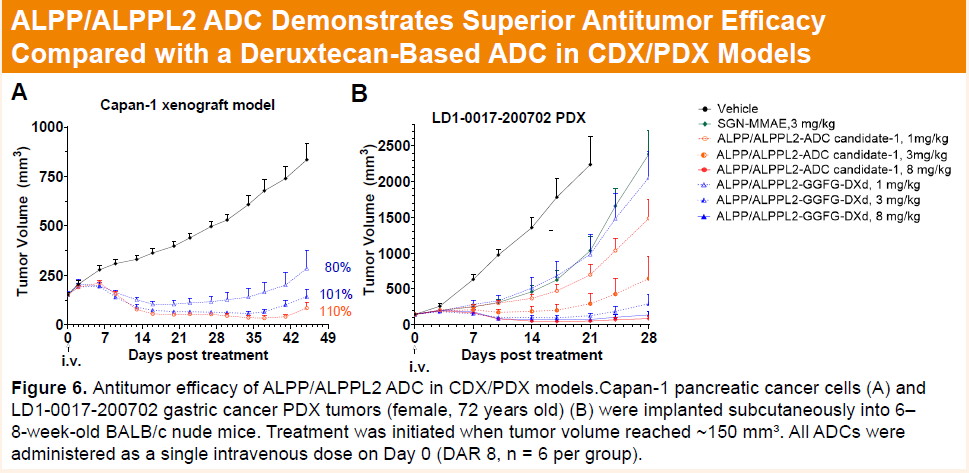
HLX85 demonstrated a more than 40-fold enhancement in bystander killing effect compared with deruxtecan-based ADCs, which may help overcome tumor heterogeneity. In vivo, a single dose of 1 mg/kg HLX85 induced marked tumor regression in the Capan-1 CDX model. In a gastric adenocarcinoma PDX model refractory to MMAE-based ADCs, a single 8 mg/kg dose achieved deep tumor remission.
-
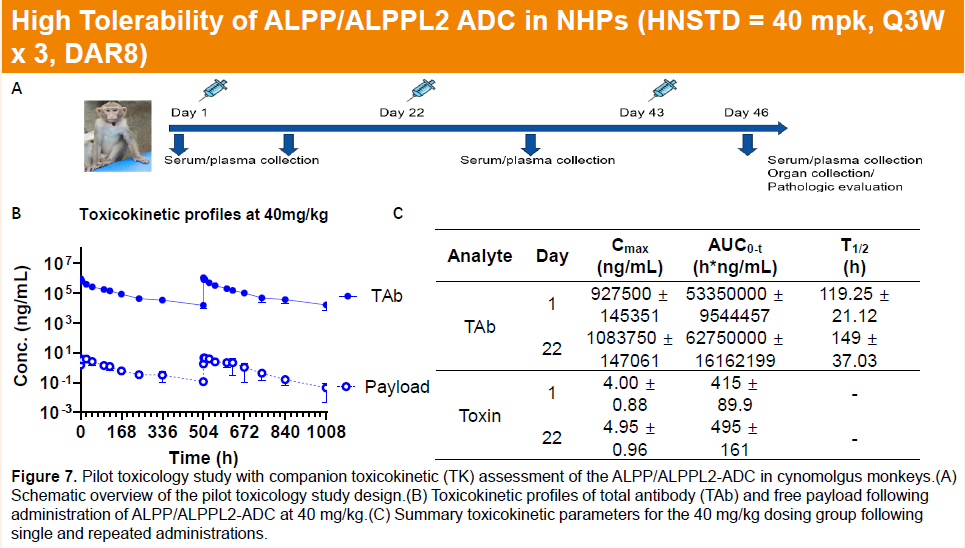
In non-human primates, HLX85 exhibited linear pharmacokinetics and favorable tolerability, with a highest non-severely toxic dose (HNSTD) of 40 mg/kg.

Overall, the data presented at AACR 2026 highlight the differentiated design and broad potential of the Hanjugator™ platform, as well as the promising therapeutic profiles of multiple ADC candidates, supporting further clinical development across a range of solid tumor indications, including potential combination strategies with immuno-oncology (IO) therapies to further enhance anti-tumor activity.
Looking forward, Henlius has established a diversified innovation ecosystem spanning multispecific TCEs, antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The company remains committed to addressing unmet clinical needs and accelerating the translation of innovative assets into clinical value, with the goal of delivering high-quality and accessible therapies to patients worldwide.
