Shanghai, China, April 22, 2026 — Shanghai Henlius Biotech, Inc. (2696.HK) today announced the first presentation of preclinical data for its novel CD3×CD28×STEAP1 trispecific T cell engager (TCE), HLX3902, at the 2026 Annual Meeting of the American Association for Cancer Research (AACR) in a poster session. Henlius continues to expand its proprietary TCE platform into multiple “immune desert” solid tumors. To date, the platform has generated more than five early-stage innovative assets.
Among them, HLX3901 (DLL3×DLL3×CD3×CD28 tetraspecific antibody) and HLX3902 (STEAP1×CD3×CD28 trispecific antibody) represent key pipeline candidates targeting solid tumors including small cell lung cancer (SCLC) and prostate cancer, respectively, and have advanced to the IND approval/acceptance stage. Notably, HLX3901 has demonstrated potential best-in-class therapeutic profile in preclinical studies, further validating the platform’s productivity and scalability in solid tumor drug discovery.
Proprietary Multispecific TCE Platform Incorporating CD28 Co-stimulation to Remodel “Immune Desert” Solid Tumors— Enhanced efficacy with an expanded therapeutic window
Limited T cell infiltration and an immunosuppressive tumor microenvironment (TME) remain major barriers to the efficacy of TCE therapies in solid tumors. To address these challenges, Henlius has developed a multispecific TCE platform incorporating CD28 co-stimulatory signaling. By delivering both signal 1 (CD3-mediated activation) and signal 2 (CD28 co-stimulation), the platform enables targeted T cell recruitment and robust activation, enhancing T cell proliferation, persistence, and cytotoxic activity within suppressive TMEs.
The platform incorporates multiple engineering strategies to optimize safety and efficacy, including target-dependent CD3 activation to minimize off-target T cell activation, conditional CD28 co-stimulation that is triggered only upon CD3 engagement, and a cis-binding design of the CD3 and CD28 domains to reduce nonspecific T cell activation and fratricide. Together, these features contribute to an expanded therapeutic window and an improved safety profile.
AACR 2026: HLX3902 Demonstrates Potent and Durable Antitumor Responses in Prostate Cancer
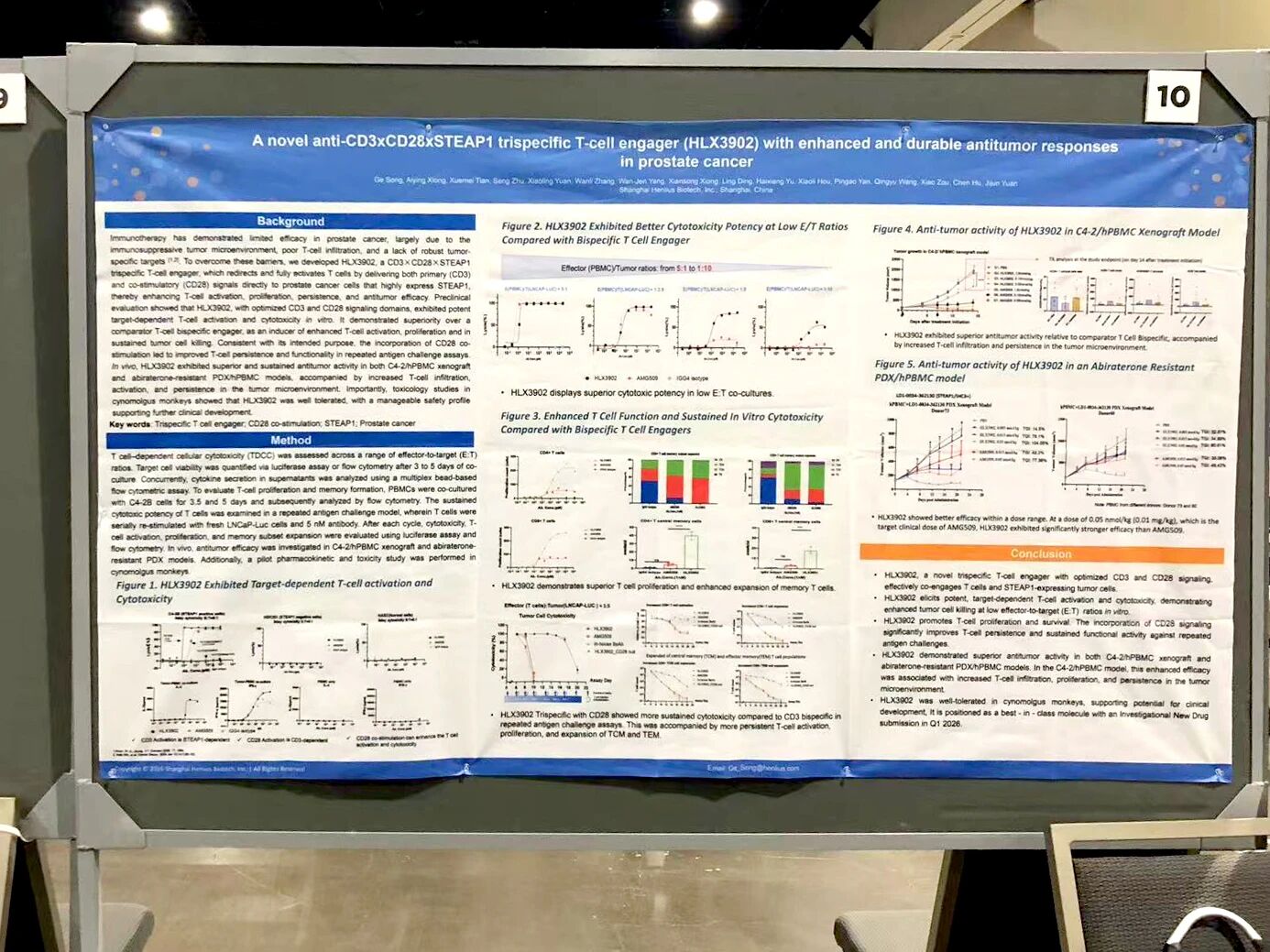
STEAP1 is highly expressed in prostate cancer and represents an attractive tumor-specific target. HLX3902, a CD3×CD28×STEAP1 trispecific TCE, enables dual T cell activation while selectively targeting STEAP1-expressing tumor cells. Its IND application has been accepted by the Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA).
Key preclinical findings:
-
In vitro, HLX3902 induced target-dependent T cell activation and cytotoxicity
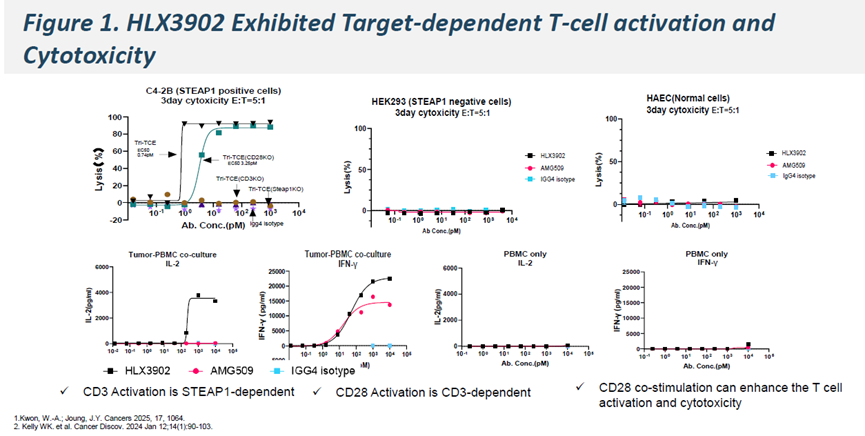
-
Compared with CD3-only bispecific TCEs, HLX3902 demonstrated enhanced cytotoxicity at low E:T ratios (1:5–1:10)
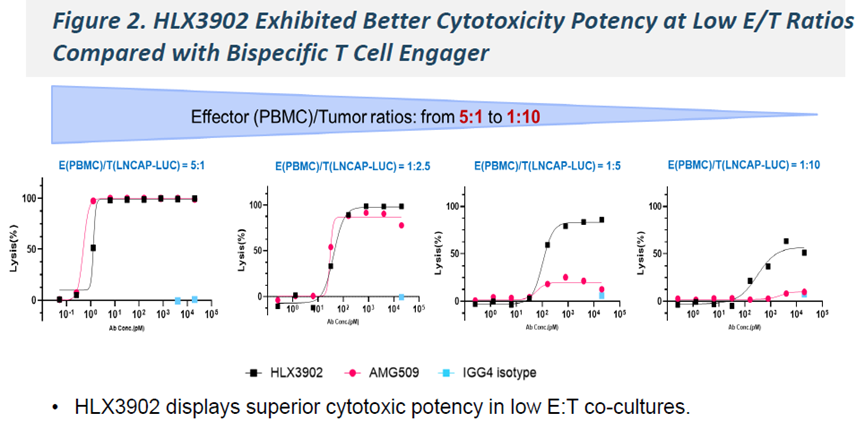
-
In repeated antigen stimulation models, HLX3902 demonstrated more sustained cytotoxic activity, along with enhanced T cell activation, proliferation, and expansion, and an increased proportion of memory T cell populations, including both central memory (TCM) and effector memory (TEM) T cells.
-
In vivo, HLX3902 demonstrated superior antitumor activity compared with bispecific TCEs in C4-2/hPBMC xenograft models, with enhanced efficacy associated with increased T cell infiltration within the tumor microenvironment, as well as improved T cell activation and persistence.
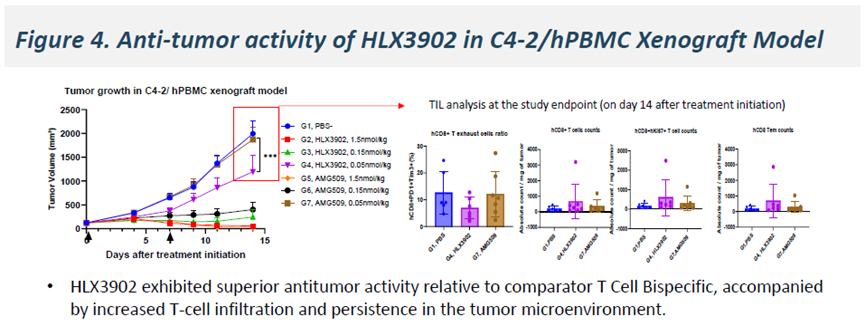
-
In abiraterone-resistant PDX/hPBMC models, HLX3902 also showed superior antitumor efficacy versus AMG509 at 0.05 nmol/kg (0.01 mg/kg), a dose within AMG509’s reported clinical target range.
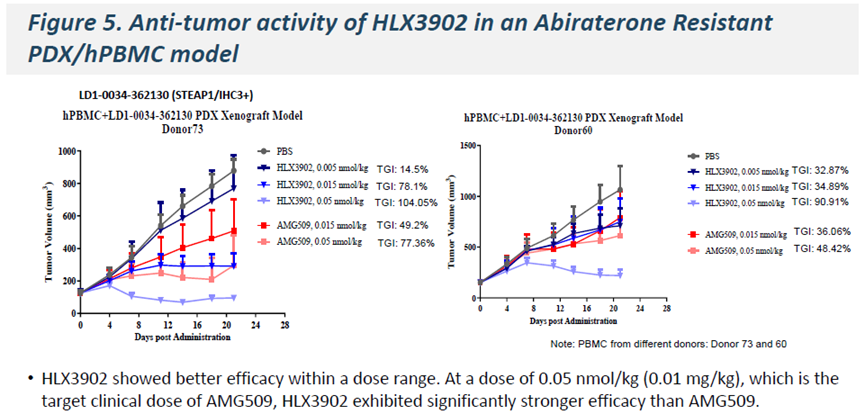
-
In a preliminary pharmacokinetic and toxicology study in cynomolgus monkeys, HLX3902 was well tolerated with a manageable safety profile, supporting its further clinical development.
AET 2025: HLX3901 – AI-Designed Next-Generation TCE for Solid Tumors
AET 2025 marked the first disclosure of HLX3901, a DLL3×DLL3×CD3×CD28 tetraspecific TCE and a key asset derived from Henlius’ proprietary platform. The IND application for HLX3901 was approved by the NMPA in March 2026. By integrating AI-driven epitope discovery with multispecific TCE engineering, HLX3901 is designed to achieve durable and highly specific T cell activation, overcome the challenge of low T cell infiltration in solid tumors, and reduce the risk of cytokine release syndrome (CRS).
-
HLX3901 employs a dual-epitope targeting strategy against DLL3, enabling simultaneous binding to two distinct epitopes to enhance binding avidity and improve target engagement and pharmacological activity.
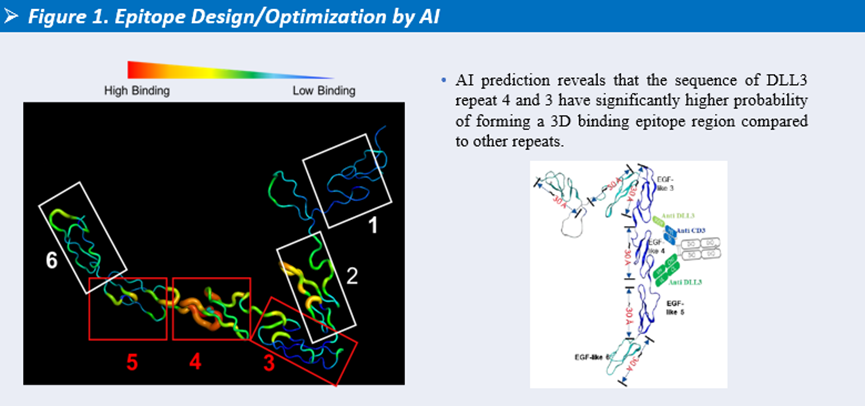
-
During epitope discovery, Henlius used AI-based prediction to identify DLL3 antibody-binding regions, selecting Repeat 3 and Repeat 4 as the primary epitope sites. Based on this, a dual-epitope antibody targeting EGF-like 3 and EGF-like 4 was designed. In vitro studies showed that this design achieved improved cytotoxic activity across different effector-to-target ratios.
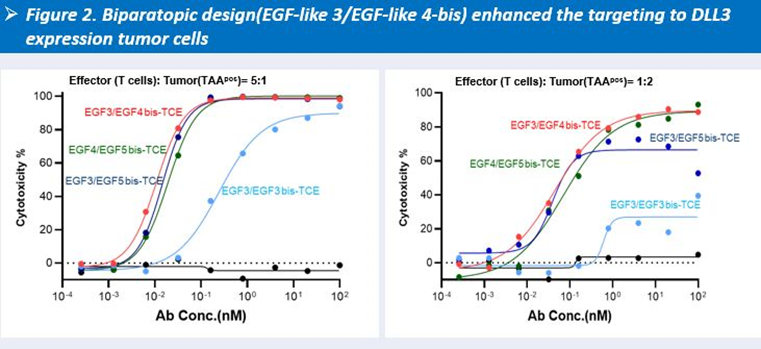
-
HLX3901 incorporates an Fc-silenced design that extends systemic half-life while reducing Fc-mediated nonspecific immune activation.
-
In preclinical studies, HLX3901 demonstrated superior cytotoxicity compared with Tarlatamab, particularly at low effector-to-target ratios, along with upregulation of Bcl-xL and expansion of memory CD8⁺ T cell populations in vitro.
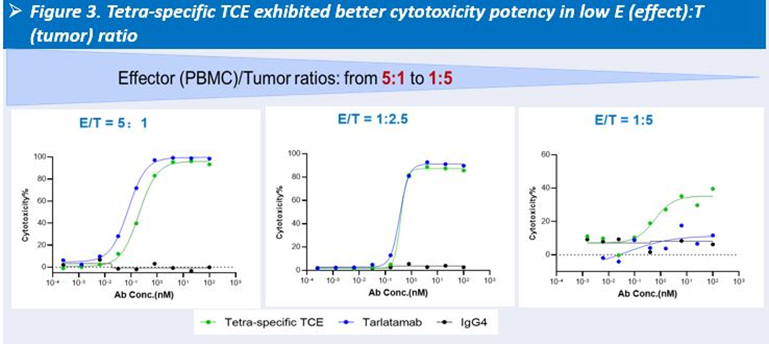
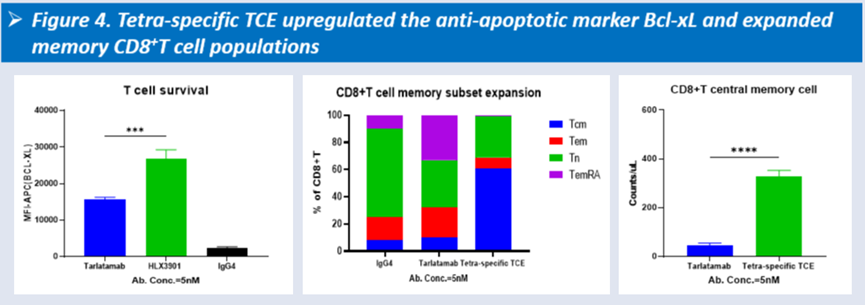
-
In vivo, it showed dose-dependent tumor growth inhibition in SHP77 CDX models, with stronger and more durable antitumor efficacy than Tarlatamab, accompanied by enhanced T cell infiltration within the tumor microenvironment.
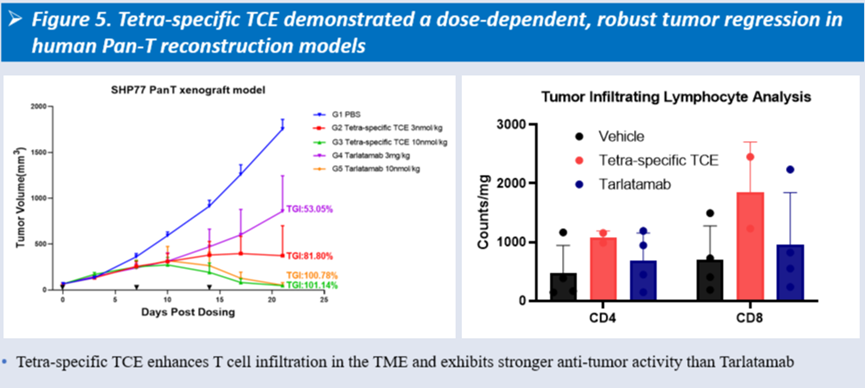
-
From a safety perspective, HLX3901 exhibited reduced non-specific CD4⁺ and CD8⁺ T cell activation and lower cytokine release (including IL-2, IFN-γ, TNF-α, and IL-6) compared with Tarlatamab. In cynomolgus monkeys, it was well tolerated with a no-observed-adverse-effect level (NOAEL) of 10 mg/kg, suggesting a wider therapeutic window.
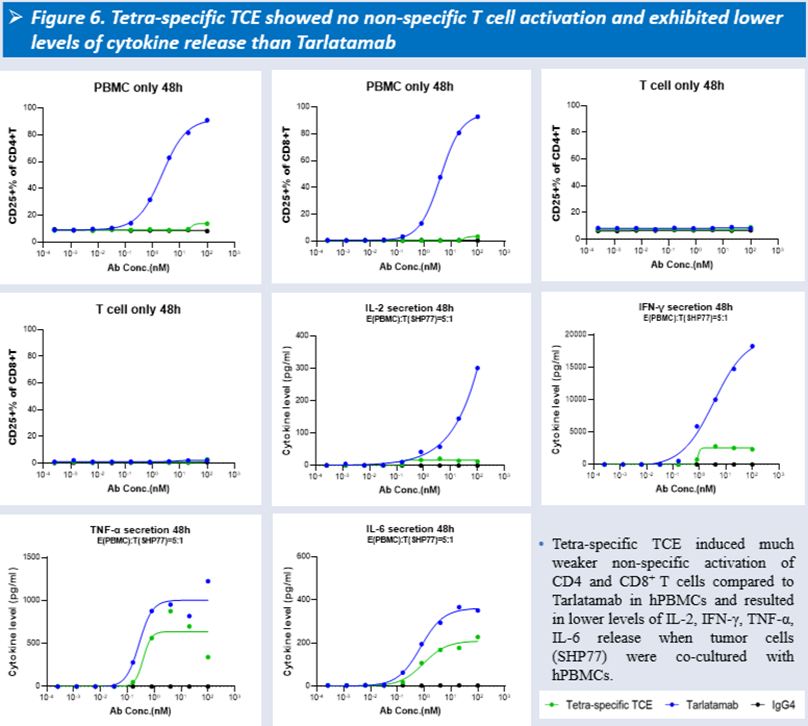
Beyond monotherapy potential, HLX3901 also shows promise for combination strategies within Henlius’ immuno-oncology(IO) portfolio. In combination with the anti–PD-1 antibody serplulimab (trade name: Hetronifly® in Europe), it may enhance tumor-directed T cell killing while relieving PD-1/PD-L1-mediated T cell exhaustion, thereby potentially achieving synergistic antitumor activity.
Looking forward, Henlius has established a diversified innovation ecosystem spanning multispecific TCEs, antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The company remains committed to addressing unmet clinical needs and accelerating the translation of innovative assets into clinical value, with the goal of delivering high-quality and accessible therapies to patients worldwide.
